|
X) Radius: It is of the order of 10 -15 cm. Viii) Charge on one mole of electron is approx. Vii) Mass of one mole of electrons: It is 0.55 mg. 
Iv) Specific charge: e/m ratio is called specific charge and was determined by Thomson as 1.76x108 coulombs/gm. (nearly equal to 1/1837th of mass of hydrogen atom). Iii) Mass: It was found by J.J.Thomson as 9.11x10 -28 g. Ii) Charge: It was determined by Millikan by oil drop experiment as -1.602x10 -19 coulombs or 4.803x10 -10 e.s.u. Thomson in 1886, through the study of cathode rays (Julius Plucker) and the name electron was proposed by Stoney. In this relative mass calculation we have assume that a neutron has a mass of 1, then the relative masses of proton and electron are.įundamental Properties of Proton neutron and Electronġ) Electron: Electron is a universal constituent of matter. Relative Mass of Proton Neutron and electron are Therefor when we calculate amu the mass of electron is negated. Still if we compare the mass of protons, neutron and electron it is observed that mass of Neutrons and Protons are almost equal whereas mass of electron are very less as compared to mass of proton and Neutrons. 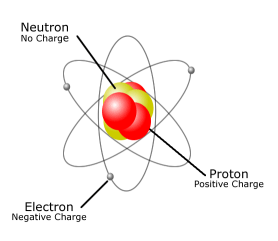 
Sub atomic particles are extremely small and almost mass less. Relative masses of protons, neutrons and electrons
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
